If you ever wondered how you can determine the elements that make something up, you can do that by subjecting your sample to spectral analysis. This involves breaking it down into its spectrum to determine its chemical composition and properties. Spectral analysis is commonly used to find out the composition of unknown materials and utilize instruments called spectroscopes. Spectroscopes can be generally defined as instruments that disperse electromagnetic radiation or spectra.
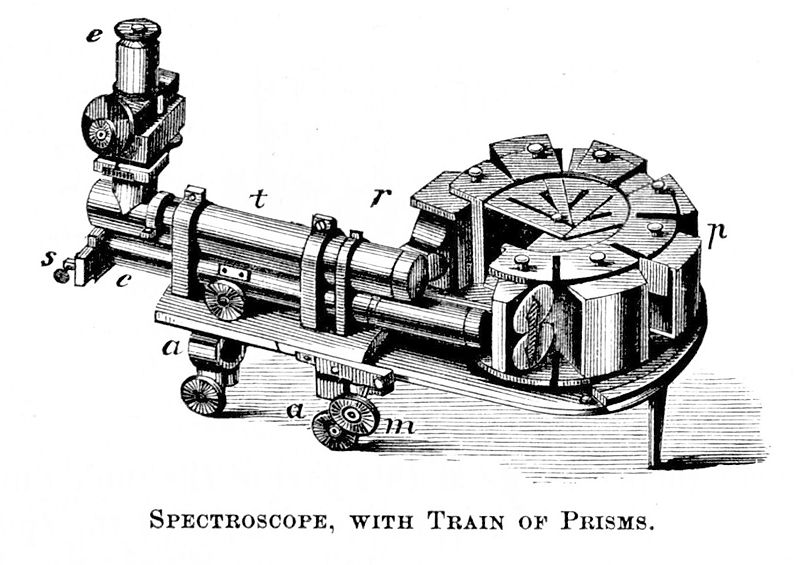
Modern spectroscopes break down the light source into its spectrum through a diffraction grating. This is similar to splitting the light by passing it through a slit and allowing it to display its components. Knowing the intensity of the light at different wavelengths allows scientists to determine what makes it up, or its chemical composition and even the rate by which the light is moving.
This has many practical applications in astronomy since we are able to determine the composition of distant stars without having to travel thousands of miles just to find out what they are made of. Through the use of spectroscopes, the light from a star under study can be focused and then split up. The corresponding spectrographs can then be analyzed to determine which elements make up that particular star. Those trained to read such graphs immediately know a star or material’s chemical composition by the intensity of the light at a particular wavelength. For example, one can know if sodium is present by looking at its two particular bright bands at the 588-589 nanometer wavelengths.
